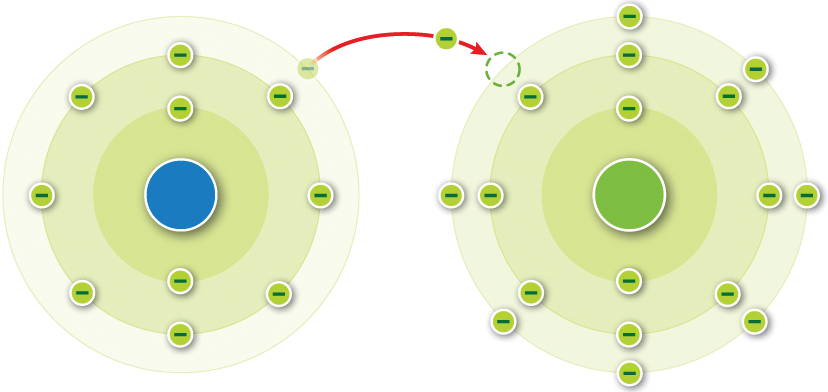  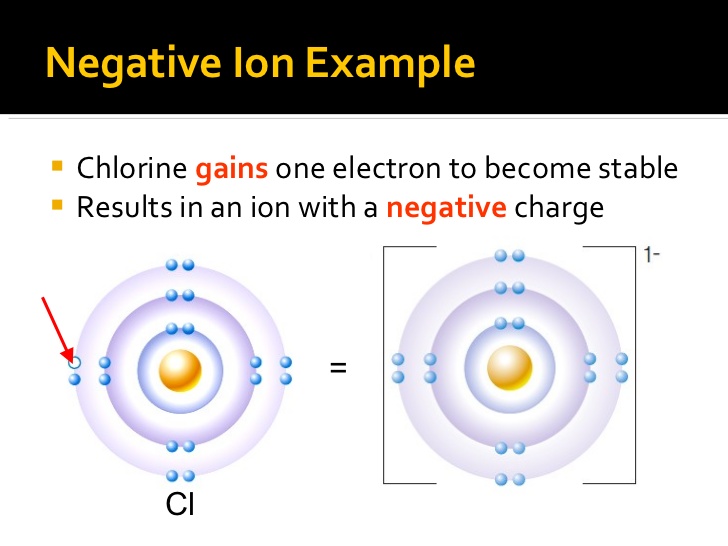
The process of ionization will be discussed more in depth in the X-rays module. The amount of ionization that occurs is dependent upon two main factors, (1) the radiation energy, and (2) the type of material the radiation is interacting with. They are formed by the loss of electrons. Cations are positive, they have more protons than electrons. There are broadly two types of ions: cations and anions. If they do, they form charged particles called ions. But atoms are capable of gaining and losing electrons. In order to liberate an electron from its parent atom, it will take energy that is at least equal to the binding energy that holds the electron. They have the same number of protons and electrons. Negatively charged ions are called anions. An ion with extra electrons has a negative charge and is. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. An ion is an atom with a different number of electrons than protons and is electrically charged. This means that the electrons are held in their electron shells by a quantity of binding energy. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. One way ionization occurs is through radiation.ÂĪtoms are held together by a binding energy. If it has more electrons than protons it has a net negative charge and is known. Consequently, the liberated electron is a negative ion, as long as it exists by itself and does not combine with another atom. If it has a net charge, (more or less electrons than protons) it is an ion. With an electron removed, the atom possesses a plus one charge, therefore it is a positive ion. That is, there are more protons in the nucleus (positive charges) than there are electrons (negative charges). If we remove an electron from a stable atom, the atom becomes electrically incomplete/unbalanced. Ionization is any process that changes the electrical balance within an atom. After reading this section you will be able to:Īn ion is an atom, group of atoms, or a particle with a positive or negative charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
